2026-01-14 – Press Releases – www.prnewswire.com
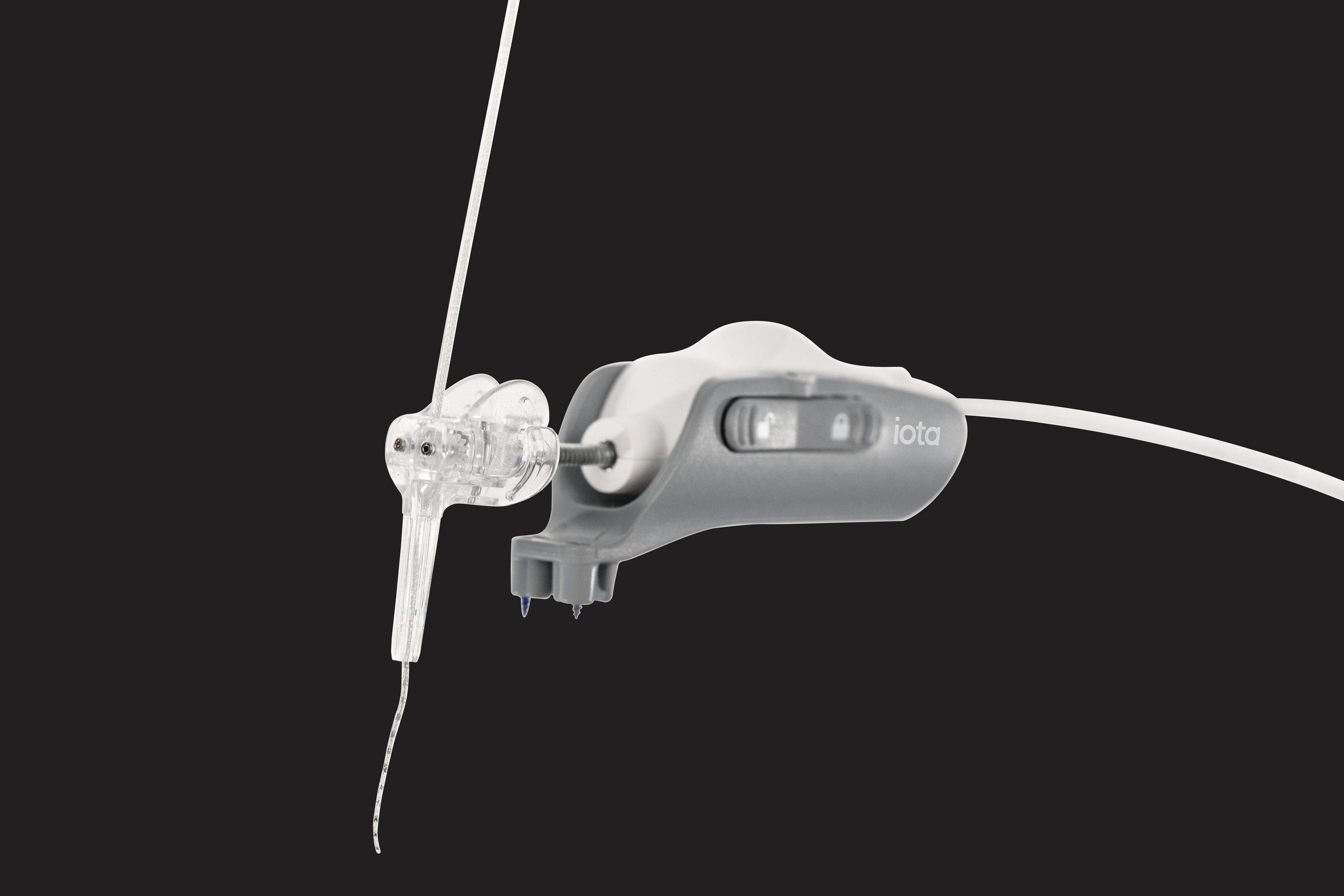
ST. PAUL, Minn., Jan. 14, 2026 /PRNewswire/ — iotaMotion, Inc., creator of iotaSOFT®, the first and only FDA cleared robotic-assisted cochlear implant insertion system, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for expanded pediatric use of its iotaSOFT®…